|
ATOMIC AND PHYSICAL PROPERTIES OF THE GROUP 2 ELEMENTS This page explores the trends in some atomic and physical properties of the Group 2 elements - beryllium, magnesium, calcium, strontium and barium. You will find separate sections below covering the trends in atomic radius, first ionisation energy, electronegativity and physical properties. Even if you aren't currently interested in all these things, it would probably pay you to read most of this page. The same ideas tend to recur throughout the atomic properties, and you may find that earlier explanations help to you understand later ones. The physical properties are extremely difficult to explain, however. Trends in Atomic Radius | |||||||
|
Note: You will find atomic radius covered in detail in another part of this site. If you choose to follow this link, use the BACK button on your browser to return quickly to this page. | |||||||
|
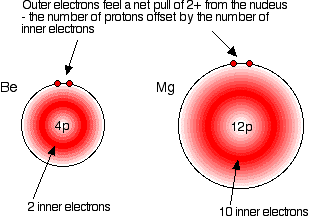
You can see that the atomic radius increases as you go down the Group. Notice that beryllium has a particularly small atom compared with the rest of the Group. Explaining the increase in atomic radius The radius of an atom is governed by
Compare beryllium and magnesium:
| |||||||
|
Note: If you aren't sure about writing electronic structures using s and p notation it might be a good idea to follow this link before you go on. Use the BACK button on your browser to return quickly to this page. | |||||||
|
In each case, the two outer electrons feel a net pull of 2+ from the nucleus. The positive charge on the nucleus is cut down by the negativeness of the inner electrons.
This is equally true for all the other atoms in Group 2. Work it out for calcium if you aren't convinced. The only factor which is going to affect the size of the atom is therefore the number of layers of inner electrons which have to be fitted in around the atom. Obviously, the more layers of electrons you have, the more space they will take up - electrons repel each other. That means that the atoms are bound to get bigger as you go down the Group. | |||||||
|
Note: You may think that this is all a bit long-winded! It is, after all, fairly obvious that atoms will get bigger if you add more layers of electrons. Why, then, bother about exploring the net pull on the electrons from the centre of the atom? It is a matter of setting up good habits. If you are talking about atoms in the same Group, the net pull from the centre will always be the same - and you could ignore it without creating problems. That isn't true if you try to compare atoms from different parts of the Periodic Table. If you don't get into the habit of thinking about all the possible factors, you are going to make mistakes. | |||||||
|
Trends in First Ionisation Energy First ionisation energy is the energy needed to remove the most loosely held electron from each of one mole of gaseous atoms to make one mole of singly charged gaseous ions - in other words, for 1 mole of this process:
| |||||||
|
Note: You will find ionisation energy covered in detail in another part of this site. If you choose to follow this link, use the BACK button on your browser to return quickly to this page. | |||||||
|
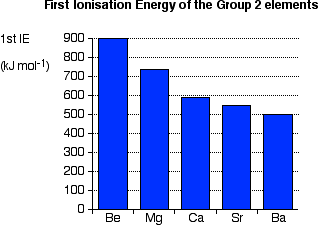
Notice that first ionisation energy falls as you go down the group. Explaining the decrease in first ionisation energy Ionisation energy is governed by
As you go down the Group, the increase in nuclear charge is exactly offset by the increase in the number of inner electrons. Just as when we were talking about atomic radius further up this page, in each of the elements in this Group, the outer electrons feel a net attraction of 2+ from the centre. However, as you go down the Group, the distance between the nucleus and the outer electrons increases and so they become easier to remove - the ionisation energy falls. Trends in Electronegativity Electronegativity is a measure of the tendency of an atom to attract a bonding pair of electrons. It is usually measured on the Pauling scale, on which the most electronegative element (fluorine) is given an electronegativity of 4.0. | |||||||
|
Note: You will find electronegativity covered in detail in another part of this site. If you choose to follow this link, use the BACK button on your browser to return quickly to this page. | |||||||
|
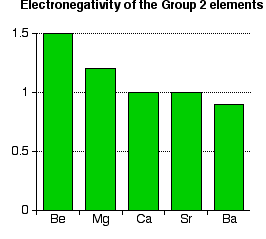
All of these elements have a low electronegativity. (Remember that the most electronegative element, fluorine, has an electronegativity of 4.0.) Notice that electronegativity falls as you go down the Group. The atoms become less and less good at attracting bonding pairs of electrons. | |||||||
|
Note: You might argue that the fall doesn't apply throughout the Group because both calcium and strontium appear to have an electronegativity of 1.0. This is probably most easily explained by the fact that electronegativities are often only recorded to 1 decimal place. To two decimal places, calcium is 1.00 and strontium is 0.95. When these numbers are rounded to 1 decimal place, both would appear to have an electronegativity of 1.0. | |||||||
|
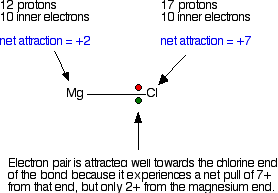
Explaining the decrease in electronegativity Imagine a bond between a magnesium atom and a chlorine atom. Think of it to start with as a covalent bond - a pair of shared electrons. The electron pair will be dragged towards the chlorine end because there is a much greater net pull from the chlorine nucleus than from the magnesium one.
The electron pair ends up so close to the chlorine that there is essentially a transfer of an electron to the chlorine - ions are formed. The large pull from the chlorine nucleus is why chlorine is much more electronegative than magnesium is. Now compare this with the beryllium-chlorine bond. The net pull from each end of the bond is the same as before, but you have to remember that the beryllium atom is smaller than a magnesium atom. That means that the electron pair is going to be closer to the net 2+ charge from the beryllium end, and so more strongly attracted to it.
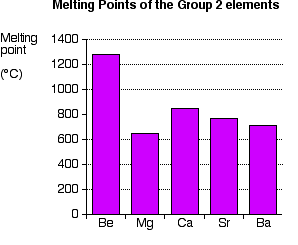
In this case, the electron pair doesn't get attracted close enough to the chlorine for an ionic bond to be formed. Because of its small size, beryllium forms covalent bonds, not ionic ones. The attraction between the beryllium nucleus and a bonding pair is always too great for ions to be formed. Summarising the trend down the Group As the metal atoms get bigger, any bonding pair gets further and further away from the metal nucleus, and so is less strongly attracted towards it. In other words, as you go down the Group, the elements become less electronegative. As you go down the Group, the bonds formed between these elements and other things such as chlorine become more and more ionic. The bonding pair is increasingly attracted away from the Group 2 element towards the chlorine (or whatever). Trends in Melting Point, Boiling Point, and Atomisation Energy The facts Melting points
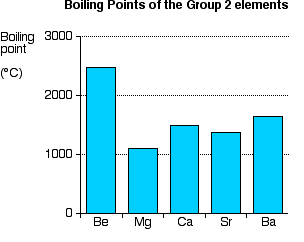
You will see that (apart from where the smooth trend is broken by magnesium) the melting point falls as you go down the Group. Boiling points
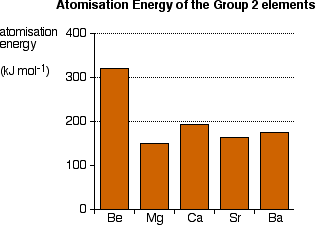
You will see that there is no obvious pattern in boiling points. It would be quite wrong to suggest that there is any trend here whatsoever. Atomisation energy This is the energy needed to produce 1 mole of separated atoms in the gas state starting from the element in its standard state (the state you would expect it to be in at approximately room temperature and pressure).
And again there is no simple pattern. It looks similar to, but not exactly the same as, the boiling point chart. Trying to explain this (up-date May 2020) The only explanations you are ever likely to meet relate to the melting points, and any simple explanation you come across is likely to be wrong. A recent email discussion with a university lecturer in general and inorganic chemistry suggests that the problem may be even deeper than I had imagined, and I no longer have the confidence to discuss this in any detail. There is one book that I know about which is honest enough to admit the difficulty. A.G.Sharpe, in his degree level book Inorganic Chemistry admits that there is no easy explanation for the variations in the physical data in Group 2. If that is indeed the case, as looks pretty likely, then it is much better at this level to have no explanation than a deeply flawed one.
© Jim Clark 2002 (last modified December 2021) |
|||||||