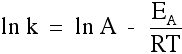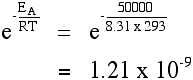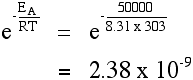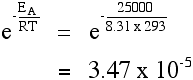|
RATE CONSTANTS AND THE ARRHENIUS EQUATION This page looks at the way that rate constants vary with temperature and activation energy as shown by the Arrhenius equation. | ||
|
Note: If you aren't sure what a rate constant is, you should read the page about orders of reaction before you go on. This present page is at the hard end of the rates of reaction work on this site. If you aren't reasonably confident about the basic rates of reaction work, explore the rates of reaction menu first. | ||
|
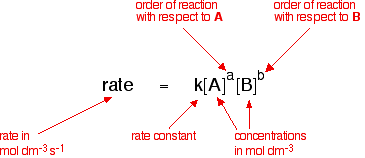
The Arrhenius equation Rate constants and rate equations You will remember that the rate equation for a reaction between two substances A and B looks like this:
| ||
|
Note: If you don't remember this, you must read the page about orders of reaction before you go on. Use the BACK button on your browser to return to this page. | ||
|
The rate equation shows the effect of changing the concentrations of the reactants on the rate of the reaction. What about all the other things (like temperature and catalysts, for example) which also change rates of reaction? Where do these fit into this equation? These are all included in the so-called rate constant - which is only actually constant if all you are changing is the concentration of the reactants. If you change the temperature or the catalyst, for example, the rate constant changes. This is shown mathematically in the Arrhenius equation. The Arrhenius equation
What the various symbols mean Starting with the easy ones . . .
| ||
|
Note: If you aren't sure about activation energy, you should read the introductory page on rates of reaction before you go on. Use the BACK button on your browser to return to this page. | ||
|
And then the rather trickier ones . . .
By this time you've probably forgotten what the original Arrhenius equation looked like! Here it is again:
You may also come across it in a different form created by a mathematical operation on the standard one:
"ln" is a form of logarithm. Don't worry about what it means. If you need to use this equation, just find the "ln" button on your calculator. Using the Arrhenius equation The effect of a change of temperature You can use the Arrhenius equation to show the effect of a change of temperature on the rate constant - and therefore on the rate of the reaction. If the rate constant doubles, for example, so also will the rate of the reaction. Look back at the rate equation at the top of this page if you aren't sure why that is. What happens if you increase the temperature by 10°C from, say, 20°C to 30°C (293 K to 303 K)? The frequency factor, A, in the equation is approximately constant for such a small temperature change. We need to look at how e-(EA / RT) changes - the fraction of molecules with energies equal to or in excess of the activation energy. Let's assume an activation energy of 50 kJ mol-1. In the equation, we have to write that as 50000 J mol-1. The value of the gas constant, R, is 8.31 J K-1 mol-1. At 20°C (293 K) the value of the fraction is:
By raising the temperature just a little bit (to 303 K), this increases:
You can see that the fraction of the molecules able to react has almost doubled by increasing the temperature by 10°C. That causes the rate of reaction to almost double. This is the value in the rule-of-thumb often used in simple rate of reaction work. | ||
|
Note: This approximation (about the rate of a reaction doubling for a 10 degree rise in temperature) only works for reactions with activation energies of about 50 kJ mol-1 fairly close to room temperature. If you can be bothered, use the equation to find out what happens if you increase the temperature from, say 1000 K to 1010 K. Work out the expression -(EA / RT) and then use the ex button on your calculator to finish the job. The rate constant goes on increasing as the temperature goes up, but the rate of increase falls off quite rapidly at higher temperatures. | ||
|
The effect of a catalyst A catalyst will provide a route for the reaction with a lower activation energy. Suppose in the presence of a catalyst that the activation energy falls to 25 kJ mol-1. Redoing the calculation at 293 K:
If you compare that with the corresponding value where the activation energy was 50 kJ mol-1, you will see that there has been a massive increase in the fraction of the molecules which are able to react. There are almost 30000 times more molecules which can react in the presence of the catalyst compared to having no catalyst (using our assumptions about the activation energies). It's no wonder catalysts speed up reactions!
| ||
|
Note: If you read this carefully, you should notice that I am not saying that the reaction will be 30000 times faster. There may well be 30000 times more molecules which can react, but it is highly likely that the frequency factor will have changed in the presence of the catalyst. And the rate constant k is just one factor in the rate equation. You won't just have the original reactants present as before. The catalyst is bound to be involved in the slow step of the reaction, and a new rate equation will have to include a term relating to the catalyst. Nevertheless, the catalysed reaction is still going to be a lot faster than the uncatalysed one because of the huge increase in sufficiently energetic molecules. | ||
|
Other calculations involving the Arrhenius equation If you have values for the rate of reaction or for the rate constant at different temperatures, you can use these to work out the activation energy of the reaction. Only one UK A' level Exam Board expects you to be able to do these calculations. They are included in my chemistry calculations book, and I can't repeat the material on this site. | ||
|
Note: There is no way of making this sufficiently different from what is in the book to avoid being in breach of contract with my publishers if I included it on this site. If you are interested in my chemistry calculations book you might like to follow this link. | ||
To the rates of reaction menu . . . © Jim Clark 2002 (modified October 2013) |
||