|
ALUMINIUM This page starts by looking at the extraction of aluminium from its ore, bauxite, including some economic and environmental issues. It finishes by looking at some uses of aluminium. Extracting aluminium from bauxite Introduction Aluminium is too high in the electrochemical series (reactivity series) to extract it from its ore using carbon reduction. The temperatures needed are too high to be economic. Instead, it is extracted by electrolysis. The ore is first converted into pure aluminium oxide by the Bayer Process, and this is then electrolysed in solution in molten cryolite - another aluminium compound. The aluminium oxide has too high a melting point to electrolyse on its own. Aluminium ore The usual aluminium ore is bauxite. Bauxite is essentially an impure aluminium oxide. The major impurities include iron oxides, silicon dioxide and titanium dioxide. | ||||||||||||
|
Note: Bauxite actually contains one of a variety of hydrated aluminium oxides some of which you can write as Al2O3,xH2O. Since this is in itself a simplification, for UK A level purposes we normally just treat it as impure Al2O3. | ||||||||||||
|
Purifiying the aluminium oxide - the Bayer Process Reaction with sodium hydroxide solution Crushed bauxite is treated with moderately concentrated sodium hydroxide solution. The concentration, temperature and pressure used depend on the source of the bauxite and exactly what form of aluminium oxide it contains. Temperatures are typically from 140°C to 240°C; pressures can be up to about 35 atmospheres. High pressures are necessary to keep the water in the sodium hydroxide solution liquid at temperatures above 100°C. The higher the temperature, the higher the pressure needed. With hot concentrated sodium hydroxide solution, aluminium oxide reacts to give a solution of sodium tetrahydroxoaluminate.
| ||||||||||||
|
Note: You may find all sorts of other formulae given for the product from this reaction. These range from NaAlO2 (which is a dehydrated form of the one in the equation) to Na3Al(OH)6 (which is a different product altogether). What you actually get will depend on things like the temperature and the concentration of the sodium hydroxide solution. In any case, the truth is almost certainly a lot more complicated than any of these. The version I am using is perfectly acceptable and is consistent with the aluminium chemistry you will find elsewhere on the site. | ||||||||||||
|
The impurities in the bauxite remain as solids. For example, the other metal oxides present tend not to react with the sodium hydroxide solution and so remain unchanged. Some of the silicon dioxide reacts, but goes on to form a sodium aluminosilicate which precipitates out. All of these solids are separated from the sodium tetrahydroxoaluminate solution by filtration. They form a "red mud" which is just stored in huge lagoons. Precipitation of aluminium hydroxide The sodium tetrahydroxoaluminate solution is cooled, and "seeded" with some previously produced aluminium hydroxide. This provides something for the new aluminium hydroxide to precipitate around.
| ||||||||||||
|
Note: This all starts to be a bit of a nightmare if you try to get at the truth of what is happening. There are two separate issues here - both glossed over by most sources. If you like your life to be simple, ignore the rest of this note! You may well find that the product is quoted as Al2O3,3H2O. If you add everything up, that is the same as 2Al(OH)3. So which is it? I suspect that it probably isn't exactly either of these. The aluminium hydroxide originally precipitated doesn't have that formula - it's a simplification. And it will rearrange to form some sort of hydrated oxide, but I doubt whether it is as simple as the other formula suggests! The second thing is to wonder why this reaction happens at all. If you have done any aluminium chemistry, you may recognise that what normally happens is exactly the reverse of this reaction. So why is it going the other way this time? I suspect (although I don't know for sure) that it is an effect of lowering the temperature on the position of equilibrium. If the formation of the NaAl(OH)4 is endothermic, it would be favoured by high temperatures (the conditions under which the NaAl(OH)4 is formed during the first stage of the reaction). If you lowered the temperature (by cooling the reaction mixture after filtration), it would favour the exothermic change - the reverse. If you know this explanation to be wrong, please get in touch with me via the address on the about this site page. | ||||||||||||
|
Formation of pure aluminium oxide Aluminium oxide (sometimes known as alumina) is made by heating the aluminium hydroxide to a temperature of about 1100 - 1200°C.
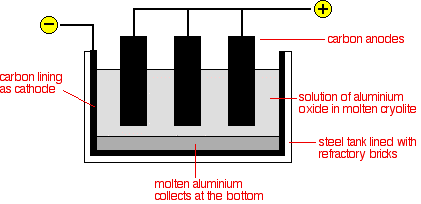
Conversion of the aluminium oxide into aluminium by electrolysis The aluminium oxide is electrolysed in solution in molten cryolite, Na3AlF6. Cryolite is another aluminium ore, but is rare and expensive, and most is now made chemically. The electrolysis cell The diagram shows a very simplified version of an electrolysis cell.
Although the carbon lining of the cell is labelled as the cathode, the effective cathode is mainly the molten aluminium that forms on the bottom of the cell. Molten aluminium is syphoned out of the cell from time to time, and new aluminium oxide added at the top. The cell operates at a low voltage of about 5 - 6 volts, but at huge currents of 100,000 amps or more. The heating effect of these large currents keeps the cell at a temperature of about 1000°C. The electrode reactions These are very complicated - in fact one source I've looked at says that they aren't fully understood. For chemistry purposes at this level, they are always simplified (to the point of being wrong! - see comment below). This is the simplification: Aluminium is released at the cathode. Aluminium ions are reduced by gaining 3 electrons.
Oxygen is produced initially at the anode.
However, at the temperature of the cell, the carbon anodes burn in this oxygen to give carbon dioxide and carbon monoxide. Continual replacement of the anodes is a major expense. | ||||||||||||
|
Note: That all seems fairly obvious, and until I was doing the research for this page, I thought it was fairly obvious too. It's a pity it turns out to be wrong! It is currently believed that neither cathode nor anode reaction happens like this. There is a reaction between the aluminium oxide and the cryolite to produce a range of complex ions involving aluminium, oxygen and/or fluorine. It is the various complexes present which gain or lose electrons at the electrodes, rearranging themselves again in the process. I'm not giving details of this because I understand that there is still some uncertainty involved. For exam purposes, you will have to use the untrue versions above because these are what are in virtually all of the textbooks at this level and are what examiners will almost certainly expect. However, if you ever meet this bit of chemistry again at a higher level, you should be aware that you will have to rethink it. | ||||||||||||
|
Some economic and environmental considerations This section is designed to give you a brief idea of the sort of economic and environmental issues involved with the extraction of aluminium. I wouldn't claim that it covers everything! | ||||||||||||
|
Note: This is deliberately brief because a lot of it is just common sense, and you will probably already have met it in detail in earlier chemistry courses, in geography, in general studies, or wherever. If you aren't sure about the various environmental problems like acid rain, global warming and the like, the very best site to find out about them is the US Environmental Protection Agency. | ||||||||||||
|
Economic considerations Think about:
Environmental problems in mining and transporting the bauxite Think about:
Extracting aluminium from the bauxite Think about:
Recycling Think about:
Uses of aluminium Aluminium is usually alloyed with other elements such as silicon, copper or magnesium. Pure aluminium isn't very strong, and alloying it adds to it strength. Aluminium is especially useful because it
Some uses include:
To the Metal Extraction menu . . . © Jim Clark 2005 (modified July 2015) |
||||||||||||