|
TESTING FOR HALIDE IONS This page describes and explains the tests for halide ions (fluoride, chloride, bromide and iodide) using silver nitrate solution followed by ammonia solution. Using silver nitrate solution Carrying out the test This test has to be done in solution. If you start from a solid, it must first be dissolved in pure water. The solution is acidified by adding dilute nitric acid. (Remember: silver nitrate + dilute nitric acid.) The nitric acid reacts with, and removes, other ions that might also give a confusing precipitate with silver nitrate. Silver nitrate solution is then added to give:
The chloride, bromide and iodide precipitates are shown in the photograph: The chloride precipitate is obviously white, but the other two aren't really very different from each other. You couldn't be sure which you had unless you compared them side-by-side. All of the precipitates change colour if they are exposed to light - taking on grey or purplish tints. The absence of a precipitate with fluoride ions doesn't prove anything unless you already know that you must have a halogen present and are simply trying to find out which one. All the absence of a precipitate shows is that you haven't got chloride, bromide or iodide ions present. The chemistry of the test The precipitates are the insoluble silver halides - silver chloride, silver bromide or silver iodide.
Silver fluoride is soluble, and so you don't get a precipitate. Confirming the precipitate using ammonia solution Carrying out the confirmation Ammonia solution is added to the precipitates.
Explaining what happens Background There is no such thing as an absolutely insoluble ionic compound. A precipitate will only form if the concentrations of the ions in solution in water exceed a certain value - different for every different compound. This value can be quoted as a solubility product. For the silver halides, the solubility product is given by the expression: Ksp = [Ag+(aq)][X-(aq)] The square brackets have their normal meaning, showing concentrations in mol dm-3. If the actual concentrations of the ions in solution produce a value less than the solubility product, you don't get a precipitate. If the product of the concentrations would exceed this value, you do get a precipitate. Essentially, the product of the ionic concentrations can never be greater than the solubility product value. Enough of the solid is precipitated so that the ionic product is lowered to the value of the solubility product. | |||||||||||||||||||
|
Note: If your syllabus says that you need to know about solubility product calculations, you might be interested in my chemistry calculations book where they are explained in detail. | |||||||||||||||||||
|
Look at the way the solubility products vary from silver chloride to silver iodide. (You can't quote a solubility product value for silver fluoride because it is too soluble. Solubility products only work with compounds which are very, very sparingly soluble.)
| |||||||||||||||||||
|
Note: These figures come from the Chemistry Data Book by Stark and Wallace. | |||||||||||||||||||
|
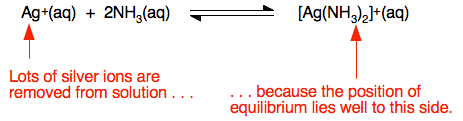
You can see that the compounds are all pretty insoluble, but become even less soluble as you go from the chloride to the bromide to the iodide. What is the ammonia doing? The ammonia combines with silver ions to produce a complex ion called the diamminesilver(I) ion, [Ag(NH3)2]+. This is a reversible reaction, but the complex is very stable, and the position of equilibrium lies well to the right.
A solution in contact with one of the silver halide precipitates will contain a very small concentration of dissolved silver ions. The effect of adding the ammonia is to lower this concentration still further. What happens if you multiply this new silver ion concentration by the halide ion concentration? If the answer is less than the solubility product, the precipitate will dissolve. That happens with the silver chloride, and with the silver bromide if concentrated ammonia is used. The more concentrated ammonia tips the equilibrium even further to the right, lowering the silver ion concentration even more. The silver iodide is so insoluble that the ammonia won't lower the silver ion concentration enough for the precipitate to dissolve. An alternative test using concentrated sulphuric acid If you add concentrated sulphuric acid to a solid sample of one of the halides you get these results:
| |||||||||||||||||||
|
Note: The chemistry of this test is explained in detail on another page. | |||||||||||||||||||
|
The only possible confusion is between a fluoride and a chloride - they would behave identically. You could distinguish between them by dissolving the original solid in water and then testing with silver nitrate solution. The chloride gives a white precipitate; the fluoride doesn't give a precipitate.
© Jim Clark 2002 (last modified March 2022) |
|||||||||||||||||||