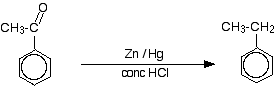|
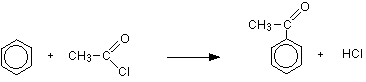
THE REACTION OF ACYL CHLORIDES WITH BENZENE This page looks at the reaction of acyl chlorides (acid chlorides) with benzene in the presence of an aluminium chloride catalyst. This is known as a Friedel-Crafts acylation. Friedel-Crafts acylation of benzene Background and equations Acylation is the term given to substituting an acyl group such as CH3CO- into another molecule. An acyl group is a hydrocarbon group attached to a carbon-oxygen double bond. For UK A level purposes, the most commonly used example of an acyl group is the ethanoyl group, CH3CO-, and so that's the one we will stick with throughout. So, if you react benzene with ethanoyl chloride in the presence of an aluminium chloride catalyst, the equation for the reaction is:
Or, simplifying it without drawing the benzene ring:
In the simplified formula for the product, the phenyl group is usually written on the left-hand side and the alkyl group to the right of the carbon-oxygen double bond - but I doubt if it really matters! The aluminium chloride isn't written into these equations because it is acting as a catalyst. If you wanted to include it, you could write AlCl3 over the top of the arrow. The product is called phenylethanone (old name, acetophenone). | ||
|
Note: If you are want the mechanism for this reaction, you will find it by following this link to another part of the site dealing with electrophilic substitution reactions. Use the BACK button on your browser to return to this page. | ||
|
Doing the reaction Ethanoyl chloride is added carefully to a mixture of benzene and solid aluminium chloride in the cold. Hydrogen chloride gas is given off. When all the ethanoyl chloride has been added, the mixture is heated under reflux at a temperature of 60°C for about 30 minutes to complete the reaction. | ||
|
Heating under reflux: Heating the flask with a condenser placed vertically in it to prevent loss of volatile liquids. Separating the product from the reaction mixture is fairly long-winded and beyond the scope of this site. You may find slight variations on the temperature and time for this reaction. | ||
|
Why the reaction is important Friedel-Crafts acylation is a very effective way of attaching a hydrocarbon-based group to a benzene ring. Although the product is a ketone (a compound containing a carbon-oxygen double bond with a hydrocarbon group either side), it is easily converted into other things. For example: The carbon-oxygen double bond can be reduced to give a secondary alcohol, which in turn can undergo a whole lot of other reactions. | ||
|
Note: This reduction is explored on a page about reduction of aldehydes and ketones. That page doesn't deal specifically with this particular ketone, but the same principles apply. Use the BACK button on your browser to return to this page. | ||
|
Phenylethanone can also be reduced to produce ethylbenzene. This is known as the Clemmensen reduction and involves heating the ketone with amalgamated zinc (a mixture of zinc and mercury) and concentrated hydrochloric acid for a long time.
This indirect route is the best way of getting an alkyl group attached to a benzene ring. It is possible to attach an alkyl group directly to the ring, but it is impossible to stop at substituting just one. An alkyl group attached to the ring makes the ring more reactive than the original benzene. That means that something like ethylbenzene reacts faster than benzene itself. The result is that you get several ethyl groups substituted around the ring rather than just one. Attaching an acyl group to the ring makes the ring so unreactive that it won't substitute a second one. | ||
|
Note: You can find more about from Friedel-Crafts reactions on a page in the arenes section, although quite of lot of it repeats what is above. Use the BACK button on your browser if you want to return to this page. | ||
© Jim Clark 2004 (modified January 2016) |
||